- Blog
- Clever fox budget planner bill organizer
- March of the phantom brigade
- Fallout new vegas new lands
- Using trackingtime
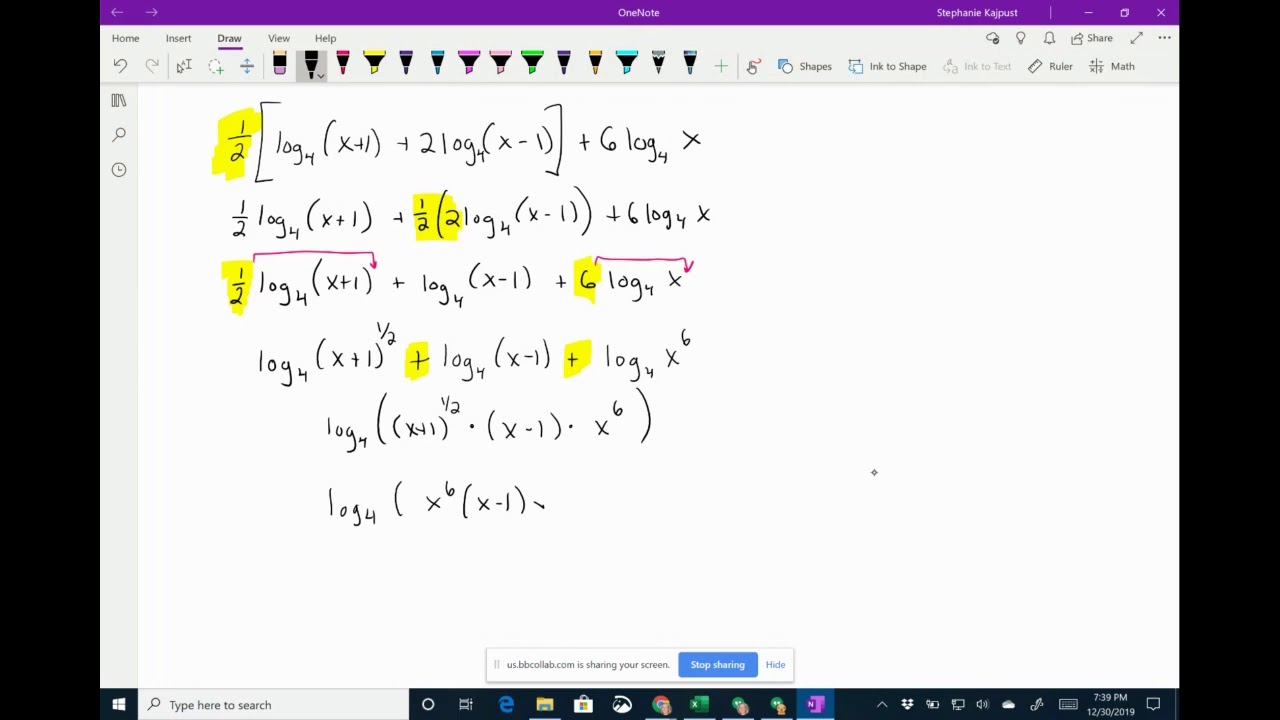
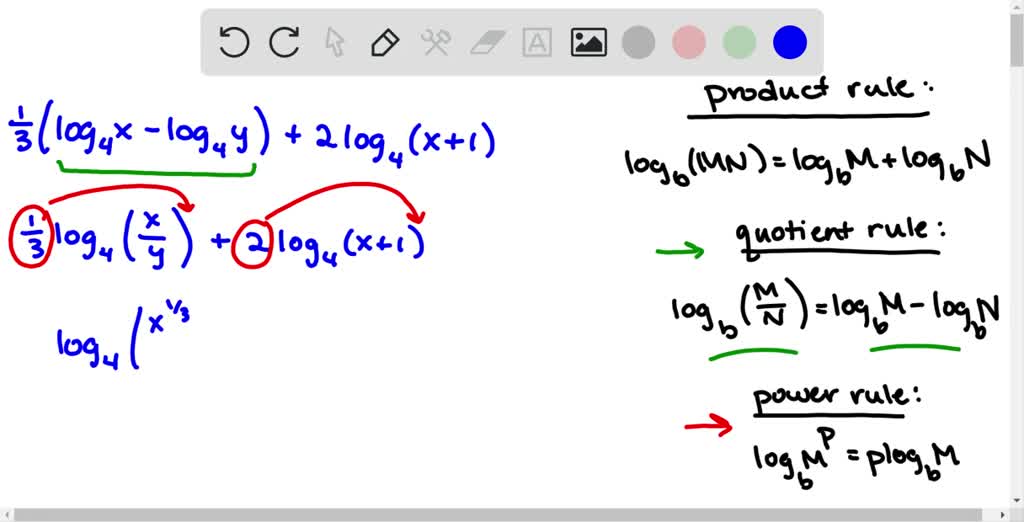
- Condense log
- Dozer rental knoxville tn
- Use of adobe illustrator
- Usps tracking package
- Motogp 21 price
- Neo and trinity
- Lzt2 task3
- Geneforge 1 mutagen map
- Grim dawn trainer riftgate unlock
- Cashflow 101
As the fluid is a liquid at this stage, the pump requires little input energy. Process 1–2: The working fluid is pumped from low to high pressure.The states are identified by numbers (in brown) in the T–s diagram. There are four processes in the Rankine cycle. Left from the bell-shaped curve is liquid, right from it is gas, and under it is saturated liquid–vapour equilibrium.

T–s diagram of a typical Rankine cycle operating between pressures of 0.06 bar and 50 bar.
However, the thermal efficiency of actual large steam power stations and large modern gas turbine stations are similar. Gas turbines, for instance, have turbine entry temperatures approaching 1500 ☌. The benefit of this is offset by the low temperatures of steam admitted to the turbine(s). By condensing the working steam vapor to a liquid the pressure at the turbine outlet is lowered and the energy required by the feed pump consumes only 1% to 3% of the turbine output power and these factors contribute to a higher efficiency for the cycle. While many substances can be used as the working fluid, water is usually chosen for its simple chemistry, relative abundance, low cost, and thermodynamic properties. Cooling towers operate as large heat exchangers by absorbing the latent heat of vaporization of the working fluid and simultaneously evaporating cooling water to the atmosphere. This 'exhaust' heat is represented by the "Q out" flowing out of the lower side of the cycle shown in the T–s diagram below. The water vapor with condensed droplets often seen billowing from power stations is created by the cooling systems (not directly from the closed-loop Rankine power cycle). Rankine engines generally operate in a closed loop where the working fluid is reused. This low steam turbine entry temperature (compared to a gas turbine) is why the Rankine (steam) cycle is often used as a bottoming cycle to recover otherwise rejected heat in combined-cycle gas turbine power stations. This gives a theoretical maximum Carnot efficiency for the turbine alone of about 63.8% compared with an actual overall thermal efficiency of less than 50% for typical power stations. Unless the pressure and temperature reach super critical levels in the boiler, the temperature range that the cycle can operate over is quite small: Steam turbine entry temperatures are typically around 565 ☌ and condenser temperatures are around 30 ☌. The efficiency of the Rankine cycle is limited by the high heat of vaporization of the working fluid. The greater the differential, the more mechanical power can be efficiently extracted out of heat energy, as per Carnot's theorem. The ability of a Rankine engine to harness energy depends on the relative temperature difference between the heat source and heat sink. Common heat sinks include ambient air above or around a facility and bodies of water such as rivers, ponds, and oceans. Possible heat sources include combustion of fossil fuels such as coal, natural gas, and oil, use of mined resources for nuclear fission, renewable fuels like biomass and ethanol, or energy capture of natural sources such as concentrated solar power and geothermal energy. The Rankine cycle closely describes the process by which steam engines commonly found in thermal power generation plants harness the thermal energy of a fuel or other heat source to generate electricity.

- Blog
- Clever fox budget planner bill organizer
- March of the phantom brigade
- Fallout new vegas new lands
- Using trackingtime
- Condense log
- Dozer rental knoxville tn
- Use of adobe illustrator
- Usps tracking package
- Motogp 21 price
- Neo and trinity
- Lzt2 task3
- Geneforge 1 mutagen map
- Grim dawn trainer riftgate unlock
- Cashflow 101
